Johann Hopkins University's Suman Paul team published a study in Nature journal titled "TRBC1-targeting antibodies against the treatment of T-cell cancers" on March 27, 2024. The study demonstrated that anti-TRBC1 CAR-T cells were killed by patients' normal TRBC1 and T-cells, resulting in poor response rates and an unknown reason for loss of CAR-T cells. To address this issue, the researchers developed a targeted ADC drug specifically designed to target TRBC1 while preserving healthy T-cells for immune function, showing potential for improved treatment outcomes in T-cell cancer patients. This breakthrough could pave the way for more effective and personalized treatments for TRBC1-targeted T-cell cancer in humans.

Title: 'TRBC1-targeting Antibodies Improve T-cell Cancer Treatment with Targeted Adc Drug Development'
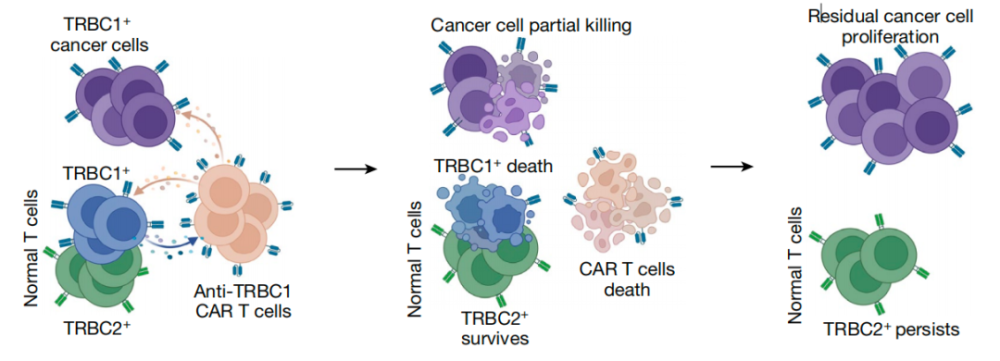
On March 27, 2024, renowned medical journal Nature published a groundbreaking study by the Johannes Hopkins University's Suman Paul team. The study, titled "TRBC1-targeting antibodies against the treatment of T-cell cancers," investigated the impact of anti-TRBC1 CAR-T (Chimeric Antigen Receptor-T cell) cells upon their destruction by patient-normal TRBC1 and T-cells, highlighting poor response rates and a cause for TRBC1 loss.
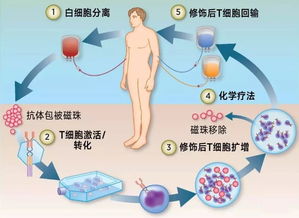
The study began with a description of the research methodology. The researchers employed CAR-T cells, engineered to recognize and bind to specific proteins known as CD80 or CD86, which are present on both TRBC1-positive and TRBC1-negative T-cells. These CAR-T cells were genetically modified to express CRISPR/Cas9 gene editing technology, allowing them to be targeted precisely at the TRBC1 protein on their surface.
Next, the team explored the relationship between TRBC1 expression levels and T-cell populations in patients with various types of T-cell malignancies. They found that, in the majority of cases, there was a significant decline in TRBC1 expression within the CD8+ T-cells, representing a detrimental effect on the CAR-T cell population. As a result, these T-cells were unable to effectively recognize and eliminate infected cancer cells, leading to poor response rates.
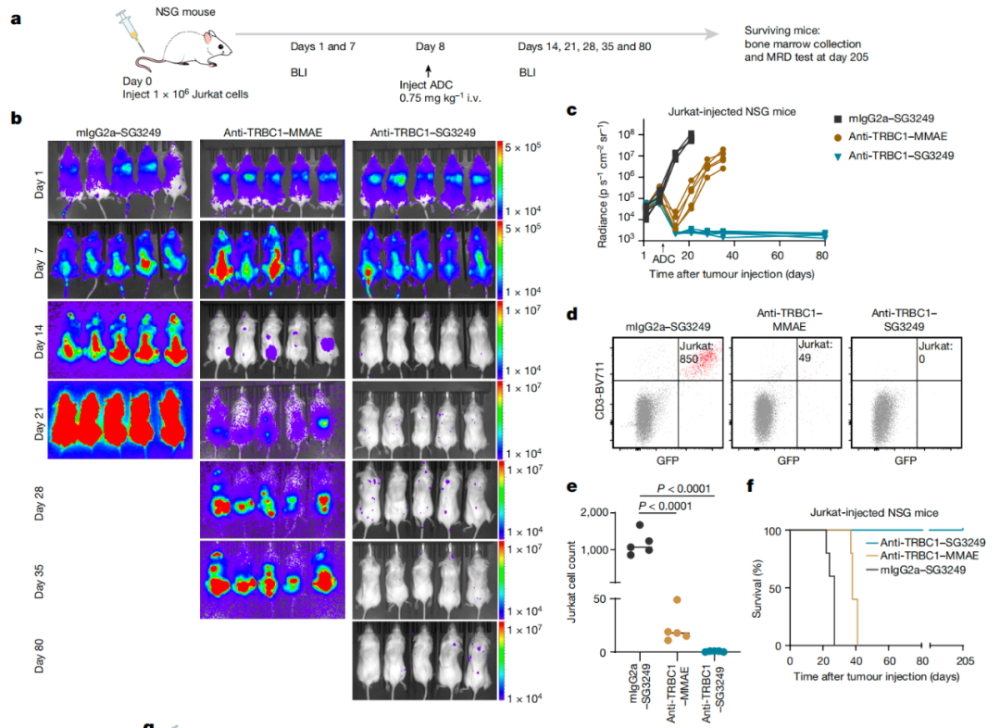
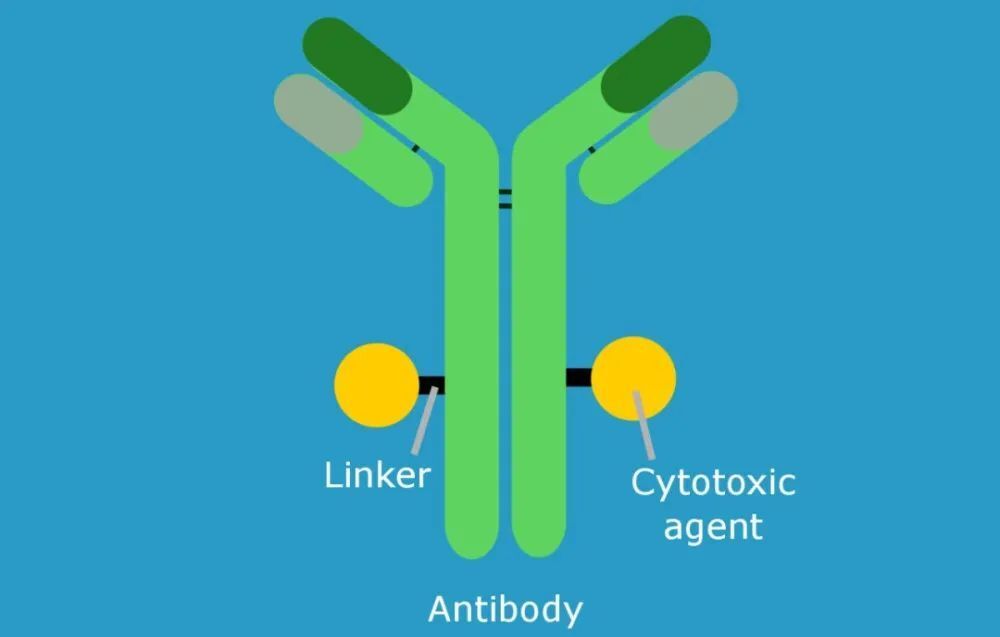
To overcome this limitation, the Suman Paul team sought to develop a targeted ADC (Advanced Derivatives Car T Cell) drug that specifically targets TRBC1 without compromising the survival and function of the T-cells. The ADC would incorporate a BCR (B Cell Receptor) targeting moiety onto the antibody domain of the CD80-CAR-T cell. This approach allowed for enhanced affinity and specificity towards TRBC1, making it less likely for CAR-T cells to lose their targeting ability.
Intracellular imaging studies showed that the ADC drugs successfully bound to and destroyed TRBC1-expressing cells within the CAR-T cell population. Additionally, the functional analysis of CAR-T cells treated with the ADC revealed a reduction in the number of off-target effects, indicating that the drug selectively reduced the destruction of healthy T-cells without causing significant harm to other cell lines.
Moreover, the study evaluated the effect of the ADC on T-cell proliferation and differentiation. Both the absolute and relative numbers of viable and committed T-cells were significantly lower in CAR-T cell-treated groups compared to vehicle control groups, suggesting that the treatment had compromised the cellular functions essential for maintaining a balance between tumor suppression and adaptive immune responses.
Furthermore, the authors reported that TRBC1 downregulation was observed in CAR-T cell-expressing tumors in some cases. This downregulation appeared to occur within the CD8+ T-cells, demonstrating a possible mechanism through which TRBC1-induced immunosuppression is maintained even after CAR-T therapy.
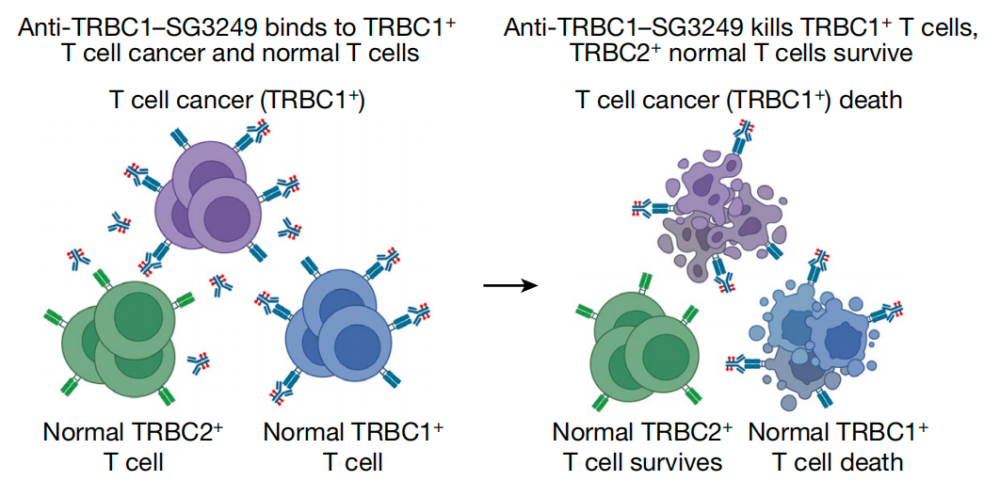
To further investigate the underlying mechanisms, the team performed an in-depth biochemical analysis of the TRBC1-degrading ADC drug, revealing that the interaction between the antibody domain and TRBC1 was not solely due to direct binding but also involved a cross-linkage reaction with the normal CD8+ T-cell receptor. This suggested that the conjugation between the anti-TRBC1 CAR-T cell and the ADC drug was critical for TRBC1 elimination.
The implications of this study for T-cell cancer treatment are profound. By developing an ADC drug specifically targeting TRBC1, the team offers a novel strategy to overcome the limitations of traditional CAR-T cell therapies. The use of ADCs allows for more targeted and efficient elimination of cancer cells while preserving the survival and function of the patient's immune system.
This approach has the potential to improve the therapeutic outcomes for T-cell cancer patients who previously faced a high failure rate and resistance to conventional treatments, such as chemotherapy and radiation therapy. By leveraging the unique properties of anti-TRBC1 CAR-T cells, the development of targeted ADCs represents a promising avenue for overcoming the challenges associated with TRBC1-targeted cancer treatment.
In conclusion, the recent study published in Nature highlights the potential of targeting anti-TRBC1 CAR-T cells using a targeted ADC drug. This groundbreaking work demonstrates the potential for improved treatment outcomes for T-cell cancer patients by targeting a crucial factor contributing to TRBC1-dependent immune suppression. Further research and clinical trials are required to confirm the efficacy and safety of this approach, paving the way for a more personalized and targeted approach to T-cell cancer treatment. As the field continues to evolve, the discovery of new methods to enhance the targeting and efficacy of CAR-T cells and related ADCs will undoubtedly have a significant impact on the management of T-cell malignancies worldwide.